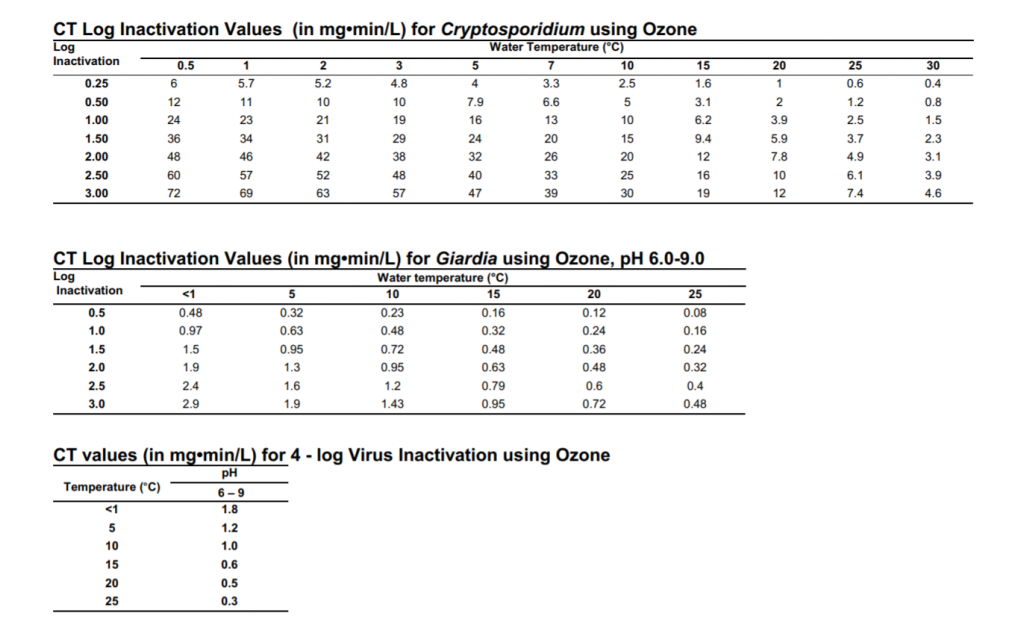
How Much Ozone Do I Need to Destroy Bacteria and Viruses?
How much ozone do I need to destroy pathogens? The question is similar to asking “how much heat do I need to cook an egg?” …
How Much Ozone Do I Need to Destroy Bacteria and Viruses? Read More »