Benzene
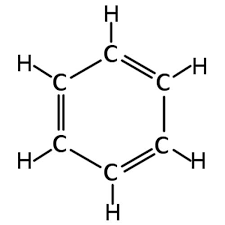
Benzene (C6H6)is a colorless flammable liquid that can be distinguished by a sweet smell resembling that of gasoline. It is released into the air when fossil fuels such as coal, petrol, and wood are burned. In fact, benzene is commonly used in unleaded fuel as a lead substitute where it allows for smoother running.
At one time, benzene concentrations in fuel could be as high as 20%. However, due to research into its potentially harmful side-effects, concentrations have been reduced to less than 1% in most countries.
Exposure to benzene can have a variety of consequences ranging from uncomfortable to potentially fatal. In high concentrations, prolonged exposure to benzene causes leukemia and can lead to the reduction of red and white blood cells from bone marrow resulting in aplastic anemia. At lower concentrations, it causes nausea, drowsiness, confusion, tremors, and headaches and can even lead to a loss of consciousness. It is irritating to the eyes and skin.
The World Health Organization (WHO) and the International Agency for Research on Cancer (IARC) classify benzene as a group one carcinogen. Because of this, no safe level of exposure can be recommended by the WHO and no specific guidelines have been recommended. EPA has sent the maximum contaminant level for benzene in drinking water at 0.005 mg/L (or 5 ppb).
Oxidation Information
Reaction with Ozone: C6H6 + 11 O3 ---> 6CO2 + 3 H2O + 11 O2
Number of O3 molecules consumed per molecule of benzene: 5.5
Molecular weights: Ozone=78.11 Benzene=48
Grams of ozone required to fully oxidize 1 gram of benzene: 3.38
Calculation: 1 gram benzene / 78.11=0.0128024581 * 48 * 5.5 moles of ozone = 3.38 grams of ozone
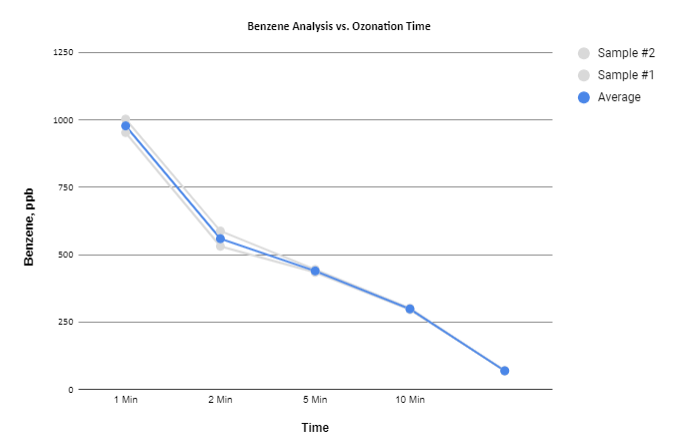
In Situ Remediation
The following chart is based on data obtained from an in-situ remediation site and displays the removal of BTEX and MBTE on two separate wells.