Well Water Treatment
Ozone Use for Well Water Treatment:
Ozone can be very effective at treating well water for residential, commercial, or agricultural applications. Ozone is also commonly used for municipal water treatment, click here for information on ozone used for municipal water treatment.
Ozone implementation can be very simple, reliable and efficient. Ozone equipment is a one-time cost with no salt, or other chemicals to buy. Only minor long-term maintenance and electrical costs are incurred. Long term, ozone can be a more cost-effective solution, while minimizing logging of salt bags and chemicals!
Common Reasons for Ozone Implementation Into Well Water Are:
-Manganese removal and filtration
-Hydrogen Sulfide (H2S) oxidation
-Water disinfection, bacteria, coliform removal
-Total Organic Carbon (TOC) removal
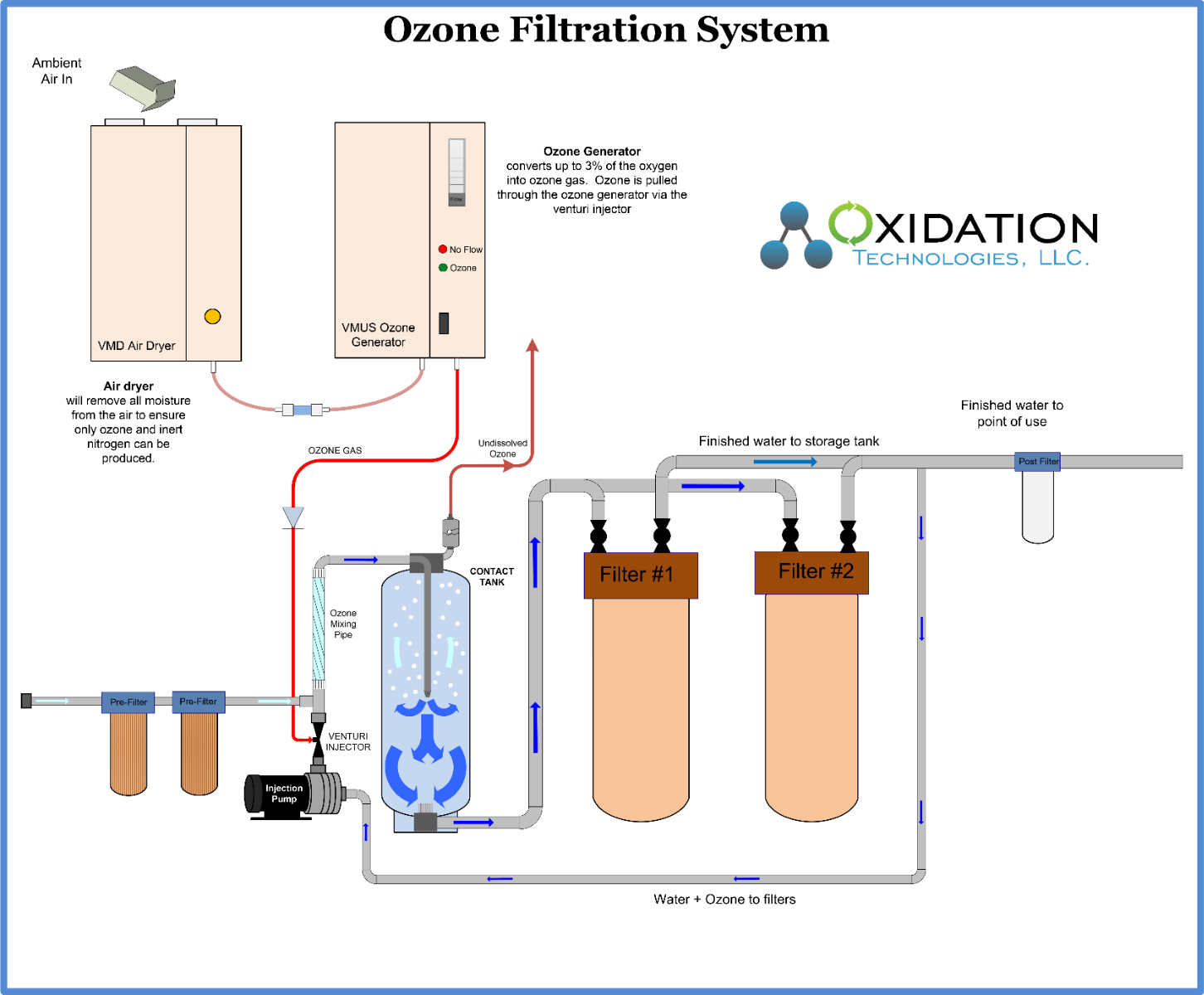
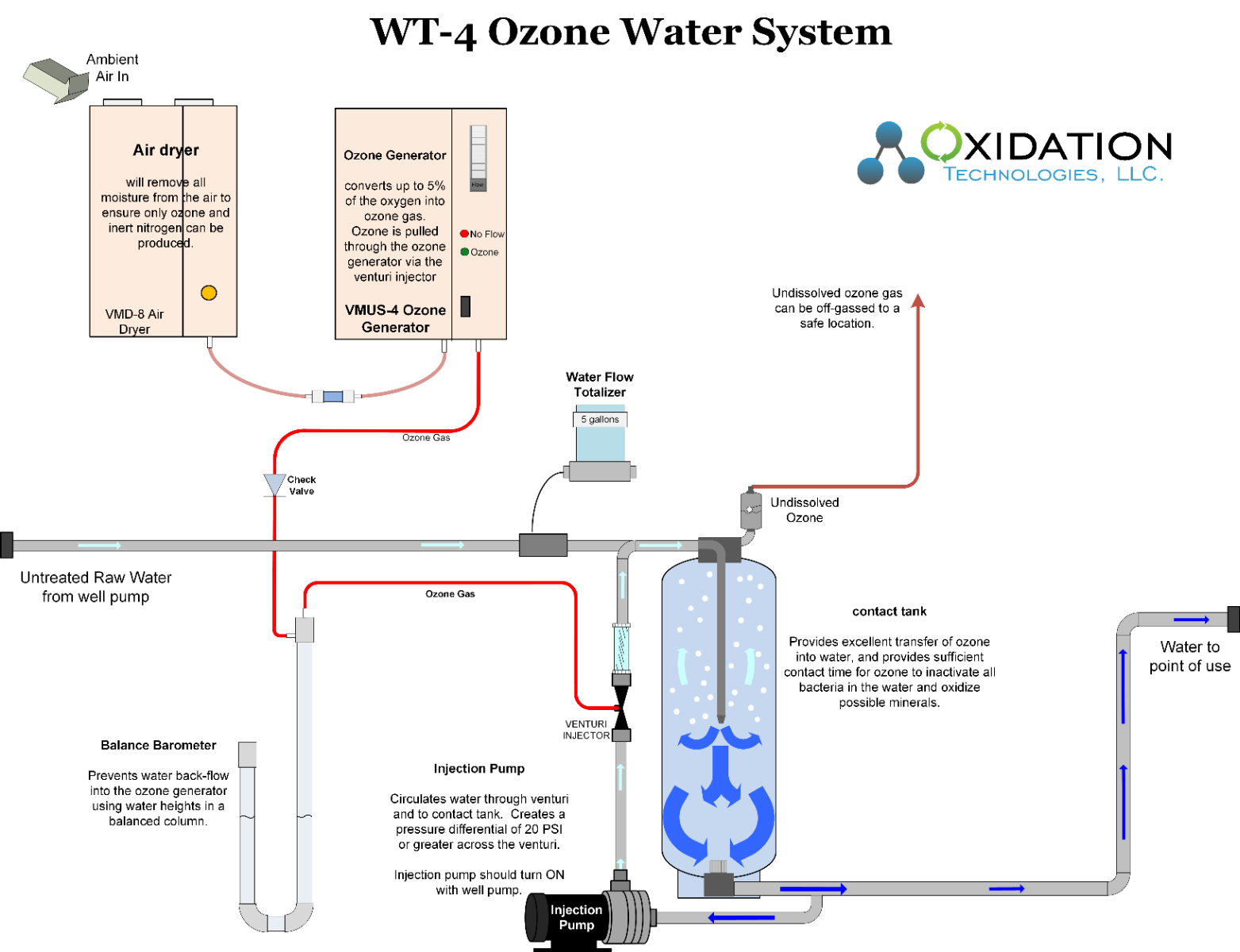
When considering ozone for well water treatment keep in mind total water consumption in a day, but also peak water demand. Remember, ozone cannot be stored and must be produced as it is used. Therefore, the ozone generator must be sized based on peak water demand, not average water flow.
Also consider when sizing an ozone system for one application, like iron removal from water, that TOC in the water, while potentially not a primary concern, will need to be considered, as this TOC will also consume ozone and create ozone demand. Be ready to provide complete water quality data when trying to determine the size of ozone system for water treatment.
*Every water is different. Also, each contaminate will require different ozone demand and may require excess contact time, or filtration to remote contaminate. Below we outline considerations when trying to determine the right ozone system for your application. This is by no means a comprehensive list, for specific recommendations for your water, contact our ozone experts today!
Iron Removal and Filtration:
Iron in water is typically found in the dissolved (Fe+2) state. In this state, iron will pass right through normal filtration but will stain appliances and fixtures over time.
Ozone is used to oxidize iron from Fe+2 to Fe+3. Iron will now bind to filter media, or anything else it touches. Using back-washable filters after the ozone contact tank will capture the now oxidized iron and remove it safely from the water.
To oxidize iron from water 0.43 ppm ozone is required for each 1 ppm of iron in the water. Therefore, multiply your iron level in water by 0.43 to establish the baseline ozone dosage rate for iron oxidation, and removal from water.
The real advantage of ozone use in iron removal from water is the complete elimination of iron bacteria. The reality is that any well water with iron, likely also carries iron bacteria. While this bacteria is not pathogenic in nature it will cause bio-film and other issues on filter beds, piping, and anywhere it can grow. When using ozone, these problems are completely eliminated.
Manganese Removal and Filtration:
Manganese in water is commonly accompanied by high levels of iron in water. Thankfully, iron and manganese both oxidize and filter from water similarly and allow for the use of the same basic system.
Manganese is found in water in the elemental Mn form. In this dissolved form manganese will flow through most filters, yet cause staining on all appliances and fixtures over time. Manganese is easily oxidized by ozone to MnO2. MnO2 can be filtered from water, as it will bind to the filter media when in the MnO2 form. Using back-washable filters after the ozone contact tank will capture the now oxidized manganese and remove it safely from the water.
To oxidize manganese from water 0.88 ppm ozone is required for each 1 ppm of manganese in the water. Therefore, multiply your manganese level in water by 0.88 to establish the baseline ozone dosage rate for manganese oxidation, and removal from water.
An important note about manganese oxidation is that if done improperly, a further oxidization state of manganese can be formed. Soluble permanganate MnO4- can be formed with excess ozone, or excess ozone contact time. It is best to use a properly designed and sized system to prevent these MnO4- from forming in your water.

Hydrogen Sulfide Oxidation:
Hydrogen Sulfide is found in well water where organics have broken down over time to form sulfides and hydrogen sulfides. H2S produces a foul odor and taste to the water. Some describe the odor as a “rotten egg” odor. Ozone can be used to efficiently, and safely remove H2S from water without the addition of any chemicals or salt.
Hydrogen Sulfide is oxidized easily by ozone into soluble Sulfite and Sulfate. Extra contact time of ozone with water will be required to ensure complete oxidation of H2S in water, however, no filtration is required for H2S removal alone.
To oxidize H2S with ozone a working dosage rate of 4 ppm of ozone for every 1 ppm of H2S has been proven effective. A contact time with water up to 10 ppm may also be required for complete removal of H2S from water. This will be dependent upon incoming H2S levels in your water.
High levels of H2S in water will also foul resin beds of a water softener. Therefore, while the softener may remove some H2S from water for a while, this will not be along term solution. Another consideration is aeration of water to remove low levels of H2S. While a low cost solution, may increase the bacteria that formed sulfide to begin with and increase overall bacteria levels in the water system. Consider using ozone prior to a water softener to reduce H2S in water safely and provide water disinfection at the same time.
Taste and Odor:
Well water and surface water may have taste and odor issues beyond the common H2S contamination. Iron, manganese, and other elemental metals in high levels can also cause taste and odor issues. Most commonly organics that are decomposing in the water source cause high bacteria, and other organics that will cause off odor or taste to your water.
Ozone will efficiently oxidize organics from water much the same as it will oxidize H2S from water. It will be helpful to ensure you have accurate water quality data to ensure ozone is an effective and complete solution to your taste and odor issues.
Water Disinfection:
Coliform bacteria or other pathogens maybe found in well water, especially when the well is shallow. These pathogens commonly

cause the well to be unused, or used for non-potable applications. Ozone can be used to kill bacteria and pathogens efficiently.
As ozone is an efficient disinfectant minimal contact time is required, therefore minimal equipment beyond the ozone generator, and ozone mixing equipment is required.
Ozone is non-selective and will destroy all pathogens in water. Unlike other chemical based solutions bacteria cannot become immune to the oxidation action of ozone. Also, using ozone will ensure residual chemicals are not added into your water supply.
Commonly when using ozone for water disinfection other benefits are realized. Ozone will enhance particulate removal and coagulation of minerals from your water, reducing the salt used in a water softener, or enhancing overall taste and odor of your water.
Total Organic Carbon (TOC) Removal:
Total Organic Carbon (TOC) is any compound containing the carbon atom that is not already fully oxidized. For example, CO2 would not be considered TOC.
TOC is found in most water sources in small amounts. High levels of TOC may be found in water containing microorganisms or other organic matter. Commonly TOC levels in surface water or shallow wells will change with the seasons, there may be spikes of high TOC levels during the spring or summer season.
Ozone can efficiently oxidize TOC into CO2 or other volatile or soluble carbonates. Only ozone in water and sufficient contact time is necessary for complete TOC removal from water. Ozone is a safe option, and a cost effective vs ongoing chemicals that are added to the water, or Activated Carbon (GAC) that will require replacement and maintenance.
Tannin Removal:
Tannins in water will cause both discoloration and odor in the water that is undesirable.
Tannin can be removed from well water using ozone with no other filtration or equipment needed. Tannins are organic matter, a form of TOC in water. Therefore, Tannin will be fully oxidized by ozone to CO, CO2, and H2O. However, Tannins may require a great deal of ozone, compared with other oxidizable compounds in water due to the complex nature of the compound. For example, Tannic Acid dissolved in water (one form of Tannin) has a molecular formula of C76H52O46. This will require both a high ozone dosage rate and a greater contact time with ozone than typical TOC in water.
Ozone dosage rates of at least 3:1 (3 mg/l ozone to 1 mg/l tannin) should be used. Ozone dosages as high as 5:1 may be necessary to eliminate color. Even with these ozone dosage rates the Tannin may not be fully oxidized to CO2, CO, and H2O, but will at least be oxidized sufficiently to remove color, and odor while leaving trace levels of generic TOC in water.
Greater ozone contact times will be required to remove Tannin in water. We offer various contact tank sizes with our well water systems. In this application, choose the largest reasonable contact tank possible.