Xylene
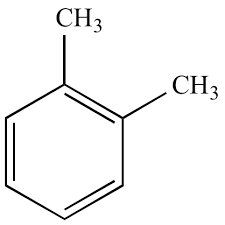
The term "Xylene" (C8H10) refers to the three isomers of dimethyl benzene: m-xylene, p-xylene, and o-xylene. The concentrations of these three xylenes are generally added together to be measured as total xylenes. Xylene is a clear, greasy liquid refined from crude oil. It is used to produce plastic bottles and polyester clothing. It can also be used as a solvent in circuit board cleaning, paint thinning, and other applications
The most common side-effect of xylene inhalation is depression of the central nervous system causing symptoms such as headaches, dizziness, nausea, and vomiting. While exposure to low concentrations of xylene does not cause permanent damage, exposure to higher concentrations can lead to symptoms such as depression, insomnia, and short-term memory loss. Xylene is an irritant to skin and strips it of its oils, making it more susceptible to permeation by other chemicals.
EPA has set the maximum contaminant level for xylene in drinking water at 10 mg/l or 10 ppm.
Oxidation Information:
Reaction with Ozone: C6H4(CH3)2 + 21 O3 ---> 8 CO2 + 5 H2O + 21 O2
Number of O3 molecules consumed per molecule of xylene: 10.5
Molecular weights: Ozone=48 xylene=106.16
Grams of ozone required to fully oxidize 1 gram of ethylbenzene: 4.75
Calculation: 1 gram ethylbenzene / 106.16 = 0.0094197438 * 48 * 10.5 moles of ozone = 4.75 grams of ozone
















