Electrolytic Ozone Production
Ozone can be produced directly in water using electrolytic ozone generators. This has huge advantages as the contacting equipment normally required for dissolving ozone gas into water is not required.
Click HERE to view Electrolytic Ozone Generators
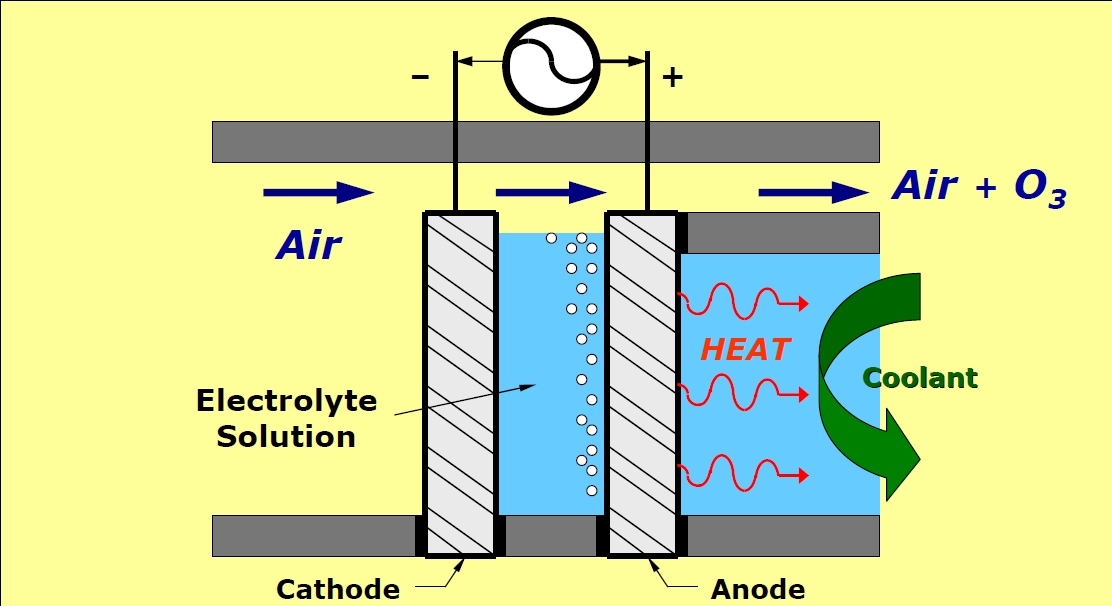
Electrolytic ozone generators use an electrical discharge in the water to split the water molecule (H2O) into H2 + O2. This O2 can also be split into O and combine to create O3. This will require a method to isolate oxygen from hydrogen and electrically charge this oxygen into ozone. Much work has been done working with catalysts, anodes and cathodes to improve efficiencies. However, this method is still unreliable in any water other than ultra-pure water, and is energy inefficient.
Advantages of Electrolytic Ozone Generator:
Ozone produced directly in water, no ozone contacting equipment or off-gassing equipment required
Compact design and size
Disadvantages of Electrolytic Ozone Generator:
High energy consumption
Short life of anode and cathode used for electrical discharge
Ozone production from Water Electrolysis:
How does an Electrolytic Ozone Generator work?
An electrolytic ozone generator (EOG) creates ozone (O3) from oxygen atoms in water (H2O). A low voltage DC current applied to water breaks the oxygen atom loose from the two hydrogen atoms. Three atoms from three water molecules join to form ozone.
Electrolytic ozone generators fall into two different types: 1) Direct Electrolysis and 2) Proton Exchange Membrane (PEM).
Direct Electrolysis: generates ozone with electrodes directly within tap water. It relies on a certain level of dissolved solids in the water to make the water conductive to the low voltage electric current. It is the simplest method and is used in equipment designed for household ozonated water needs, as well as commercial applications in dentistry, pet grooming, laundry, and sanitation for food and beverage handling equipment.
Proton Exchange Membrane (PEM): relies on a cell using ultra pure water and a membrane. The cell can produce high concentration ozone gas from this water as well as ozone dissolved in water. The ultra pure ozonated water is used in the semi-conductor industry and can be used for disinfection purposes.
Fundamentals of Ozone Production with Electrolysis
The production of ozone from water requires the input of a low voltage electrical energy to break the oxygen atoms from the hydrogen atoms in the water molecule and also an electrode with a high oxidation ability. High oxidation ability is needed to join oxygen atoms into the higher energy state of ozone (O3) instead of oxygen (O2). Electrodes made with conductive diamonds (diamond doped with boron and nitrogen) have the properties needed to join oxygen atoms into ozone. This electrode formula is resistant to breaking down and contaminating the water. Diamond electrodes suitable for ozone production are made with a Chemical Vapor Deposition (CVD) method.

In the PEM cell, a membrane is used to maintain the separation of ozone and oxygen from hydrogen after separated with electrolysis. Ultrapure water is supplied to the cell. When low voltage DC power is applied, the cell produces gaseous ozone. The water is gradually consumed as the H2O molecules break down to form ozone gas and hydrogen. A portion of the ozone dissolves into the water so that a water outlet can provide ozonated water.